System Biosciences
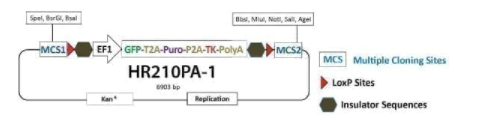
Gene Knock-Out HR Targeting Vector with TK selection [MCS1-LoxP-EF1α-GFP-T2A-Puro-P2A-hsvTK-pA-LoxP-MCS2
- SKU:
- HR210PA-1
- Availability:
- Usually Shipped in 5 Working Days
- Size:
- 10 ug
- Shipping Temperature:
- Blue Ice/ Dry Ice
Description
Gene Knock-Out HR Targeting Vector with TK selection [MCS1-LoxP-EF1α-GFP-T2A-Puro-P2A-hsvTK-pA-LoxP-MCS2]. Cat# HR210PA. Supplier: SBI System Biosciences

Overview
Get precise genomic integration of your expression cassette Use the PrecisionX™ Gene Knock-out HR Targeting Vector (MCS1-LoxP-EF1α-GFP-T2A-Puro-P2A-hsvTK-pA-LoxP-MCS2) to knock-out any gene or edit the genome. Clone your homology arms into MCS1 and MCS2, and use puromycin selection, TK selection, and/or GFP-positive imaging to find integrants. After you’ve identified clones with your gene-of-interest knocked-out or eited, you can remove the selection cassette using the Cre-LoxP system (learn more about Cre-LoxP excision here).
Why use an HR targeting vector?
Even though gene knock-outs can result from DSBs caused by Cas9 alone, SBI recommends the use of HR targeting vectors (also called HR donor vectors) for more efficient and precise mutation. HR donors can supply elements for positive or negative selection ensuring easier identification of successful mutation events. In addition, HR donors can include up to 6-8 kb of open reading frame for gene knock-ins or tagging, and, when small mutations are included in either 5’ or 3’ homology arms, can make specific, targeted gene edits.
Choose the right HR Targeting Vector for your project

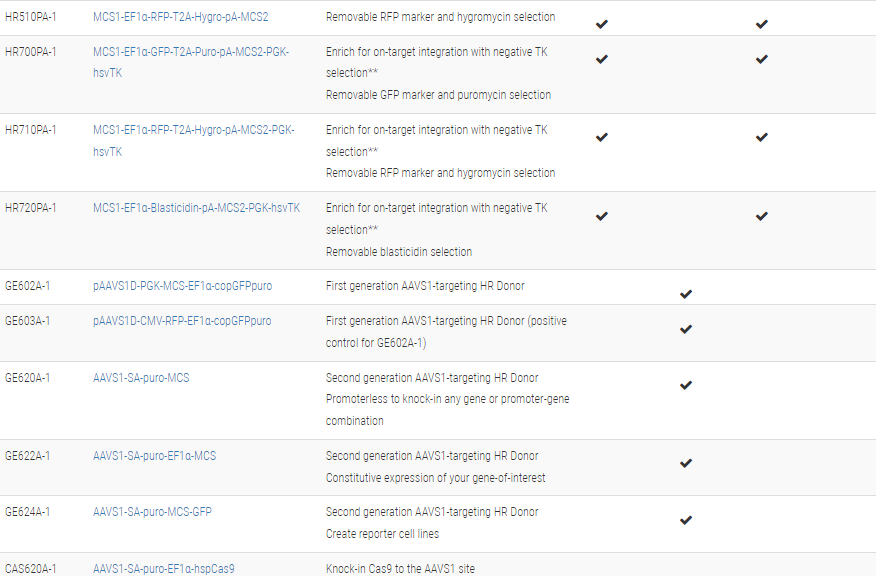

*All HR Target Vectors except PBHR100A-1 contain LoxP sites. Any sequences that are integrated between the two LoxP sites can be removed through transient expression of Cre Recombinase.
**The clever design of these HR Donors enables enrichment for on-target integration events. A PGK-hsvTK cassette is included outside of the homology arms. Because of this configuration, on-target integration that results from homologous recombination will not include the PGK-hsvTK cassette—only randomly-integrated off-target events will lead to integration of PGK-hsvTK and resulting TK activity. Therefore, TK selection will negatively select against off-target integrants. Click on any one of these vectors to see a diagram of how the negative selection works.
How It Works
At-a-glance—how to use an HR Targeting Vector to knock-out a gene
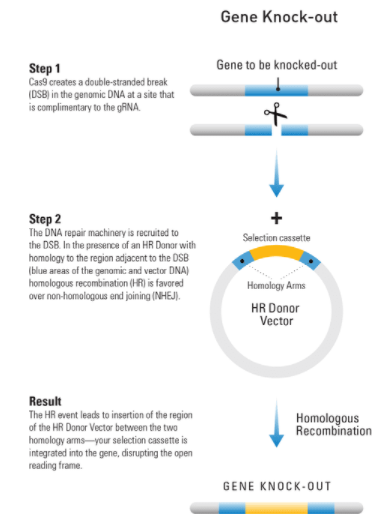
Figure 1. Knocking-out a gene using an HR Targeting Vector. Step 1: Cas9 creates a double-stranded break(DSB) in the genomic DNA at a site that is complimentary to the gRNA. Step 2: The DNA repair machinery is recruited to the DSB. In the presence of an HR Donor with homology to the region adjacent to the DSB (blue areas of the genomic and vector DNA) homologous recombination (HR) is favored over non-homologous end joining (NHEJ). Result: The HR event leads to insertion of the region of the HR Donor Vector between the two homology arms—your selection cassette is integrated into the gene, disrupting the open reading frame.
At-a-glance—how to use an HR Targeting Vector to edit a gene
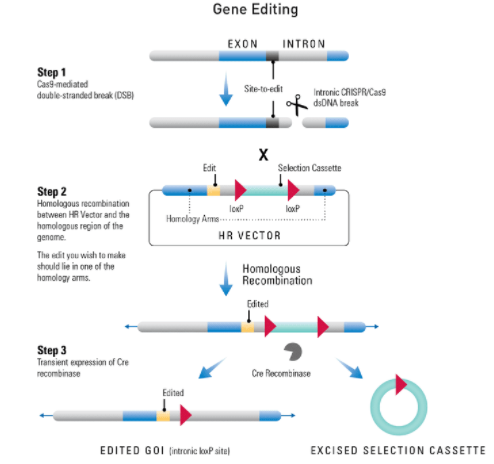
Figure 2. Editing a gene using an HR Targeting Vector. Step 1: Cas9 creates a double-stranded break (DSB) in the genomic DNA at a site that is complimentary to the gRNA. For gene editing, this DSB should be within an intron. Step 2: The DNA repair machinery is recruited to the DSB. In the presence of an HR Donor with homology to the region adjacent to the DSB (blue areas of the genomic and vector DNA) homologous recombination (HR) is favored over non-homologous end joining (NHEJ). If one of the homology arms of the HR donor contains the gene edit, it will be incorporated into the gene through the HR repair process. Step 3: Transient expression of Cre recombinase will result in excision of the selection cassette, leaving behind a single intronic LoxP site.
Genome engineering with CRISPR/Cas9
For general guidance on using CRISPR/Cas9 technology for genome engineering, including the design of HR Targeting Vectors, take a look at our CRISPR/Cas9 tutorials as well as the following application notes:
CRISPR/Cas9 Gene Knock-Out Application Note (PDF) »
CRISPR/Cas9 Gene Editing Application Note (PDF) »
CRISPR/Cas9 Gene Tagging Application Note (PDF) »







![Basic HR Targeting Vector [MCS1-LoxP-MCS2-MCS3-pA-LoxP-MCS4] for Gene Knock-In/Out Basic HR Targeting Vector [MCS1-LoxP-MCS2-MCS3-pA-LoxP-MCS4] for Gene Knock-In/Out](https://cdn11.bigcommerce.com/s-i3n9sxgjum/images/stencil/590x590/products/12477/12741/sbi%2520system%2520biosciences_1633075021__00205.original__94784.1633076533.jpg?c=1)
![Basic HR Targeting Vector [MCS1-LoxP-MCS2-MCS3-pA-LoxP-MCS4] for Gene Knock-In/Out Basic HR Targeting Vector [MCS1-LoxP-MCS2-MCS3-pA-LoxP-MCS4] for Gene Knock-In/Out](https://cdn11.bigcommerce.com/s-i3n9sxgjum/images/stencil/590x590/products/12477/15488/HR100PA-1__03435.1638779819.jpg?c=1)
![Gene Knock-Out HR Targeting Vector [MCS1-EF1α-RFP-T2A-Puro-pA-MCS2] Gene Knock-Out HR Targeting Vector [MCS1-EF1α-RFP-T2A-Puro-pA-MCS2]](https://cdn11.bigcommerce.com/s-i3n9sxgjum/images/stencil/590x590/products/12673/12938/sbi%2520system%2520biosciences_1633075021__00205.original__03121.1633123705.jpg?c=1)
![Gene Knock-Out HR Targeting Vector [MCS1-EF1α-RFP-T2A-Puro-pA-MCS2] Gene Knock-Out HR Targeting Vector [MCS1-EF1α-RFP-T2A-Puro-pA-MCS2]](https://cdn11.bigcommerce.com/s-i3n9sxgjum/images/stencil/590x590/products/12673/15643/Screenshot_2021-12-09_214631__77660.1640262373.png?c=1)
![Gene Knock-Out HR Targeting Vector [MCS1-EF1a-GFP-T2A-Puro-pA-MCS2] Gene Knock-Out HR Targeting Vector [MCS1-EF1a-GFP-T2A-Puro-pA-MCS2]](https://cdn11.bigcommerce.com/s-i3n9sxgjum/images/stencil/590x590/products/12671/12936/sbi%2520system%2520biosciences_1633075021__00205.original__02911.1633076564.jpg?c=1)
![Gene Knock-Out HR Targeting Vector [MCS1-EF1a-GFP-T2A-Puro-pA-MCS2] Gene Knock-Out HR Targeting Vector [MCS1-EF1a-GFP-T2A-Puro-pA-MCS2]](https://cdn11.bigcommerce.com/s-i3n9sxgjum/images/stencil/590x590/products/12671/15641/Screenshot_2021-12-09_212928__74392.1639067413.png?c=1)

