System Biosciences
XMIR-1 RNA oligo miRNA-1-3p with Xmotif
- SKU:
- XMIR-1
- Availability:
- Usually Shipped in 5 Working Days
- Size:
- 10 rxn
- Shipping Temperature:
- Blue Ice/ Dry Ice
Description
XMIR-1 RNA oligo miRNA-1-3p with Xmotif. Cat# XMIR. Supplier: SBI System Biosciences

Overview
Putting exosomes to work: Delivering miRNA
With SBI’s unique XMIR (and the related AXMIR) technology, you can take advantage of normal cellular processes to package a specific miRNA into exosomes, and then use those exosomes to deliver that miRNA to target cells. Choose from one of our pre-built, ready-to-transfect XMIRs (see list below). If you don’t see the miRNA you’re interested in, simply contact us at tech@systembio.com and we’ll design and synthesize the custom XMIR for you.
- Flexible—generate miRNA-packed exosomes using the cell line of your choice
- Powerful—great for a range of applications, including:
- Modulating expression of miRNA-targets in recipient cells
- Developing exosome-delivered miRNA-based therapies
How It Works
miRNA packaging into exosomes with XMIR
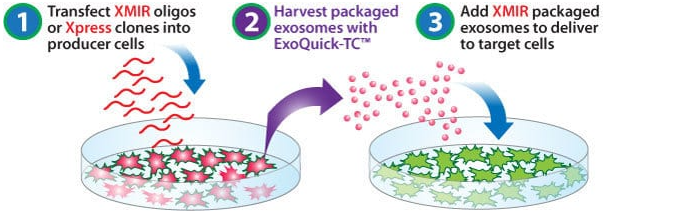
XMIR packaging relies on an RNA sequence tag—the XMotif (identified and optimized by SBI)—that targets small RNAs to exosomes for packaging. By fusing the XMotif to an miRNA oligo, you can ensure packaging of that miRNA into exosomes. Simply transfect an XMIR Oligo into exosome-producing cells, wait a short time for new exosome packaging, and isolate newly secreted exosomes containing your miRNA.
Supporting Data
XMIRs and AXMIRs can be used to modulate levels of a specific protein in target cells.
With XMIR/AXMIR technology, you can use exosomes to deliver miRNAs and anti-miRNAs to cells in order to alter levels of a specific protein (NOTE: when combined with our XStamp™ Exosome Targeting Technology, you can even generate miR/anti-miR-carrying exosomes that are targeted to a specific cell type). To demonstrate the effectiveness of using exosomes containing either miRNA or anti-miRNA to modulate protein expression levels in target cells, we generated XMIR-21 and AXMIR-21 containing exosomes from HEK293 cells, and used Western blotting to monitor expression levels of the protein PDCD4, whose expression is reduced in the presence of miR-21.
We transfected HEK293 cells with either an miR-21 XMIR Oligo (XMIR-21) or an anti-miR-21 AXMIR Oligo (AXMIR-21), isolated exosomes loaded with the respective XMIR/AXMIR, and then added these exosomes to naïve HEK-293 cells in culture. After 24 hours, total cell lysates were taken and Western blots for PDCD4, a known miR-21 target, were performed (Figure 1A), with GAPDH protein levels used as a loading control and reference signal for band intensity quantitation analysis. Quantitation is shown in Figure 1B.
These data clearly show that XMIR and AXMIR-containing exosomes can modulate expression of the target protein, as addition of exosomes from cells transfected with XMIR-21 resulted in down regulation of endogenous levels of PDCD4, and addition of exosomes from cells transfected with AXMIR-21 resulted in increased levels of PDCD4. These results also confirm that XMIRs act on endogenous targets and that AXMIRs effectively act as miRNA inhibitors in cells when delivered via exosomes.
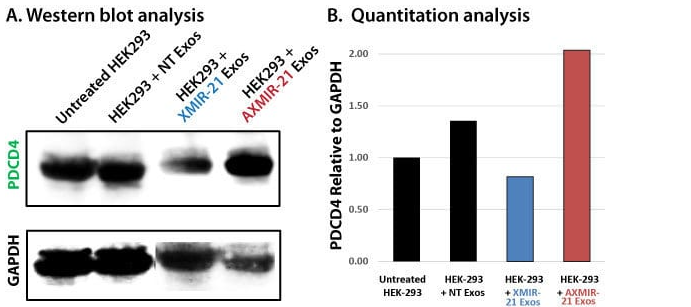
Figure 1. XMIR-21 knockdown and AXMIR-21 upregulation of the miR-21 target PDCD4. (A) Western blot data and (B) quantitation.














