System Biosciences
ExoQuick PLUS Exosome Purification Kit - for serum and plasma
- SKU:
- EQPL10A-1
- Availability:
- Usually Shipped in 5 Working Days
- Size:
- 10 reactions
- Shipping Temperature:
- Blue Ice
Description
ExoQuick PLUS Exosome Purification Kit - for serum and plasma. Cat# EQPL10A. Supplier: SBI System Biosciences

Overview
We have recently released our most advanced EV isolation system to-date—ExoQuick ULTRA—unmatched combination of yield, purity, ease of use, and cost. Learn more.
Quick and efficient exosome isolation for sensitive applications
While the ExoQuick® family of reagents speed-up and simplify exosome isolation and are compatible with most downstream uses, some applications are especially sensitive to carry-over of non-exosomal proteins—applications such as mass spectrometry, Western blotting of low abundance markers, exosome labeling, and in vivo /ex vivo exosome delivery. Which is why SBI offers ExoQuick PLUS.
Less is more—ExoQuick PLUS and ExoQuick-TC PLUS for when you need an extra level of confidence:
- Better—delivers as much as 150-fold higher yields than ultracentrifugation-based methods
- Cleaner—dramatically reduces the amount of carry-over protein compared to other column- or precipitation-based kits, and even ultracentrifugation-based methods
- Powerful—detects biomarkers with as much as a 3-fold increase in sensitivity
- Fast—takes less than 10 minutes of hands-on-time after initial ExoQuick isolation
Building on our popular ExoQuick family of products, ExoQuick PLUS delivers dramatically reduced albumin contamination in serum/plasma preparations compared to ultracentrifugation-based isolation methods (Supporting Data, Figure 1), and considerably less carryover protein compared to other exosome isolation kits (Supporting Data, Figure 2). Even better, isolation with the ExoQuick PLUS family of products results in much higher yields than isolation with ultracentrifugation—as much as 150-fold higher (Supporting Data, Figure 3).
With a simple treatment of our ready-to-use, customized ExoQuick PLUS Microspheres (kits contain enough ExoQuick and pre-packed Microspheres for 10 reactions*), you can obtain highly pure, intact exosomes suitable for a variety of protein-sensitive applications. Total hands-on time after ExoQuick isolation is less than 10 minutes!
*For exosome isolation from serum or plasma, one reaction is defined as 250 µL of serum or plasma. For exosome isolation from tissue culture media, one reaction is defined as 5 mL of media.

Learn more about ExoQuick—click here.
Choose the right ExoQuick for your application:

How It Works
High-throughput, quantitative exosome recovery
ExoQuick PLUS can be used to purify exosomes from plasma1, serum2, and malignant ascites3. With a simple workflow involving minimal hands-on time and low input sample volume requirements, ExoQuick PLUS is an excellent option for researchers who need to purify multiple exosome samples and/or samples from small animal models or clinical research samples that will then be used in sensitive downstream applications.
To isolate exosomes from cleared serum, plasma, or ascites fluid, simply:
- Add an appropriate volume of ExoQuick PLUS to as little as 100 µl sample
- Incubate for at least one hour at 4°C
- Pellet exosomes with a 30-minute low-speed spin (1500g)
- Resuspend pellet, add ExoQuick PLUS Microspheres, and incubate for 10 minutes at 4°C
- Remove microspheres with a 30-minute low-speed spin (1500g)
ExoQuick PLUS-isolated exosomes can be found in the supernatant.
You can verify the presence of exosomes with a number of different methods, including Western blotting for general exosome markers (CD63, CD9, CD81, and HSP70), NanoSight analysis, or EM (learn about different ways to detect exosomes and more in our Exosome Basics Guide).
REFERENCES
- Chugh PE, et al. Systemically Circulating Viral and Tumor-Derived MicroRNAs in KSHV-Associated Malignancies. PLoS Pathog. 2013. 9(7): e1003484. PMCID: PMC3715412.
- Epple LM, et al. Medulloblastoma Exosome Proteomics Yield Functional Roles for Extracellular Vesicles. PLoS ONE. 2012. 7(7): e42064. PMCID: PMC3407172.
- As featured in: Exosome Isolation for Proteomic Analyses and RNA Profiling Douglas D. Taylor, Wolfgang Zacharias and Cicek Gercel-Taylor, Serum/Plasma Proteomics, Methods in Molecular Biology, 2011, Volume 728, Part 4, 235-246.
Supporting Data
Taking efficient exosome isolation to a whole new level of quality
ExoQuick PLUS and ExoQuick-TC PLUS provide efficient exosome isolation with reduced protein carry-over, resulting in isolated exosomes suitable for most sensitive downstream applications.
ExoQuick PLUS

Figure 1. Exosomes isolated using ExoQuick PLUS show dramatically less albumin than when isolated by ultracentrifugation. Western blots of exosomes isolated from 500 µl of pooled human serum using either ExoQuick PLUS (left lane) or ultracentrifugation (right lane). 1.5 µg of total protein was loaded in each lane. The Western blot was probed with antibodies to CD9, an exosome-specific marker, and antibodies to two proteins typically found in high abundance in serum—IgG and albumin.

Figure 2. ExoQuick PLUS exosome isolation results in samples with less protein carry-over than competitors’ products. For each lane, 2.5 µg of total protein was loaded. A. Western blot probing for serum albumin, IgG, and the exosome-specific tetraspanin marker CD9 shows higher CD9:IgG and CD9:albumin ratios in the ExoQuick PLUS lane than in the competitors’ lanes. B. Ponceau staining to assess total serum protein abundance shows the least amount of carry-over protein in the ExoQuick PLUS lane.

Figure 3. Exosome isolation from serum with ExoQuick PLUS results in a higher particle:protein ratio than competitors’ products, indicating less protein carry-over. The particle:protein ratio is a measure of relative protein carry-over, with a higher ratio indicating less protein carry-over. Particle numbers were generated using NanoSight NTA analysis, and carry-over protein concentrations were measured using a BCA assay.

Figure 4. Exosomes isolated from bovine-exosome depleted media (Exo-FBS) using ExoQuick-TC PLUS show reduced protein carry-over compared to exosomes isolated using Company CGS’s product. For each lane, 5 µg of total protein was loaded. After running the gel, total protein was detected using colloidal blue staining. Data courtesy of Richard J. Jones, PhD., MD Anderson Cancer Center.

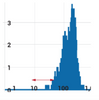
Figure 5. Exosome isolation with ExoQuick-TC PLUS results in cleaner preparations. ZetaView particle size analysis of exosomes isolated from cultured RPMI 8226 cells using either ExoQuick-TC PLUS (left panel) or a competitor’s kit (right panel). The red double arrow in the left panel indicates a broad shoulder of particles too small to be exosomes (< 50 nm in diameter) which are likely to be undesirable protein aggregates. These contaminants are not present in the exosomes isolated using ExoQuick-TC PLUS (red double arrow indicates the same particle diameter range as in the right panel). Data courtesy of Richard J. Jones, PhD., MD Anderson Cancer Center.
1 Review
-
REVIEW OF THE he ExoQuick PLUS Exosome Purification Kit for Serum and Plasma
The ExoQuick PLUS Exosome Purification Kit for Serum and Plasma is an exceptional product that has elevated my exosome research! This kit is specifically designed for isolating exosomes from complex biological fluids like serum and plasma, and it delivers impressive results.